<![CDATA[BLA Is Accepted in China for Trastuzumab Pamirtecan in HER2+ Metastatic Breast Cancer]]>
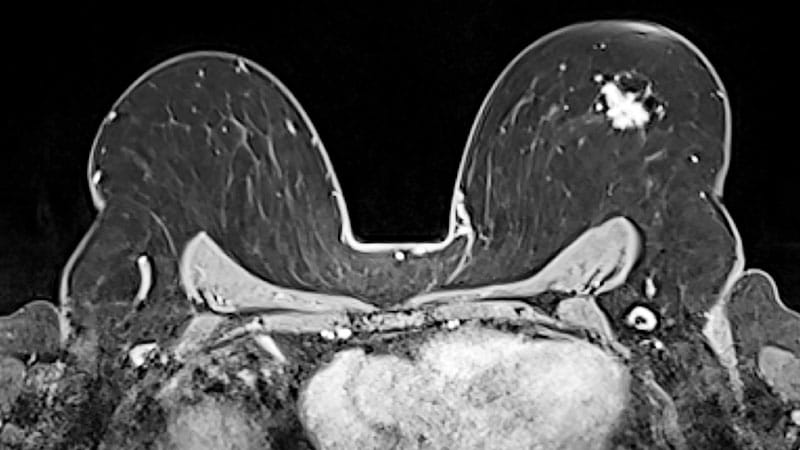
China’s National Medical Products Administration (NMPA) has accepted a biologics license application (BLA) seeking the approval of trastuzumab pamirtecan (T-Pam; DB-1303; BNT323) for the second-line treatment of adult patients with unresectable or …